Properties:
DNA & RNA: a Comparative Account:
· DNA is the genetic material in most of the organisms including many viruses, while RNA acts as the genetic material in some viruses but not in all; thus we have DNA viruses and RNA viruses. However eukaryotic cell also contain DNA in their cell organelles like Mitochondria and Plastids.
· The total concentration of DNA of a cell in a given species is constant and characteristic, but the concentration of RNA varies from one cell type to the other; active cells contain more RNA than resting cells, but the concentration can enhanced by cell activation.
· More than 90% of the cellular DNA is located in the Nucleus, whereas more than 90% of the total cellular RNA is found in cytoplasm and so also in the nucleolus as rRNA and in the rest of karyolymph as pre-mRNA-mRNA and tRNAs and few more Sc and Sn RNAs.
· A small percentage DNA is also found in cytosolic mitochondria and plastids. The amount of DNA of a gamete mother cell is exactly twice the amount of its gametes or the amount of DNA of a gamete is exactly half of the amount found in gamete mother cell.
· The DNA is a macromolecule of very high order ranging from ~ 4000 nucleotides(SS) to 4000 bp (ds) to few billion bp largest genome ever to be recorded for plants is Paris japonica with 53.2pg X 978 X 10^6, while RNA molecules, also macromolecules but they are not as big as DNA.
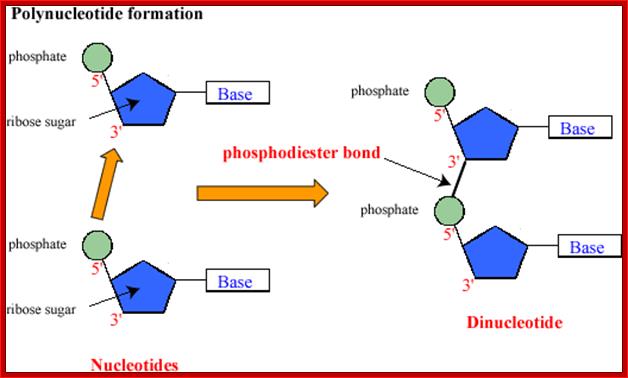
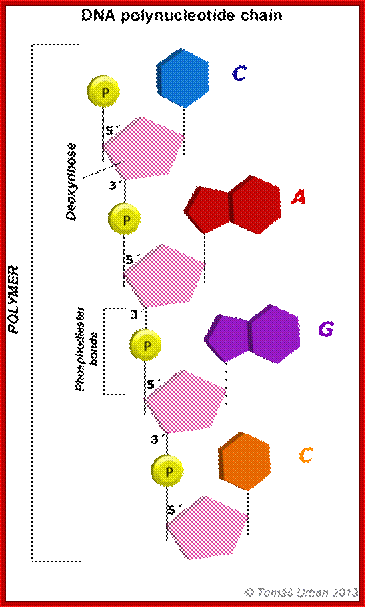
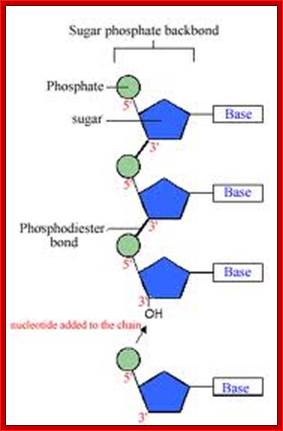
· They are polymers of nucleotides, where phosphodiester bonds covalently link nucleotides to each other.
· Both linear ssDNA have 5’OH and RNA polynucleotides have 5’ ppp their 5’ends and 3’OH both have 5’ >> 3’ polarity.
· The DNA molecules are very stable; their life span is same as that of the life span of cells, when compared to RNA whose half-life varies from few minutes to few days (rRNA 120 days).
· DNA synthesis is restricted to certain stage of cell’s life cycle, but RNA synthesis is not, it takes place all the time and goes through turnover and as long as cell exists.
· When both DNA and RNA exist in the cell, DNA acts as the genetic material and RNA plays a secondary but very important role but not as the genetic material. The RNA in viruses acts as the genetic material.
· DNA undergoes replication at a specific stage of the cell cycle, but cellular RNAs are incapable of replication.
· DNA in cellular organism is grouped into DNA-Protein structures called chromosomes, while RNAs are sparsely bound by RNPs but rRNAs with their proteins are organized into ribosomes.
· Only viral RNAs are endowed with replication ability, so also viral DNAs, but none of the other cellular RNAs have the ability to replicate. Rarely some are reverse transcribed into cDNAs; during evolution some of them transported into the nucleus and integrated into chromosomal DNA, but non functional; you can say they are ‘molecular Fossils”
· DNA molecules are very stable, but RNA molecules subjected to rapid turnover.
· Chemically DNA gives characteristic purplish color with D-amino picolinic acid (DAP) reagent; Feulgen regent, aceto-carmine and aceto orcien stains chromosomal DNA purple to reddish color while RNA gives a little greenish color reaction with Resorcinol reagent.
· DNA can exist as single stranded molecule in many viruses; it can be linear or circular depending upon the viruses. Most of the eukaryotic organisms contain DNA as ds structure and most of them organized into chromosomes and this ds DNA is linear with protected telomeric ends.
· RNA exists in the form of genetic RNA in viruses, but eukaryotic cells it plays a subsidiary role to DNA. And it is found in variety of forms such as tRNA, rRNA, mRNA, si/miRNA, snoRNA scRNA snRNA, Tasi RNA, Rasi RNA, xist RNA another 30-40 different RNAs; Only mRNAs and tRNA have coded information in the form codons and anticodon respectively.
Chemistry of Nucleic Acids:
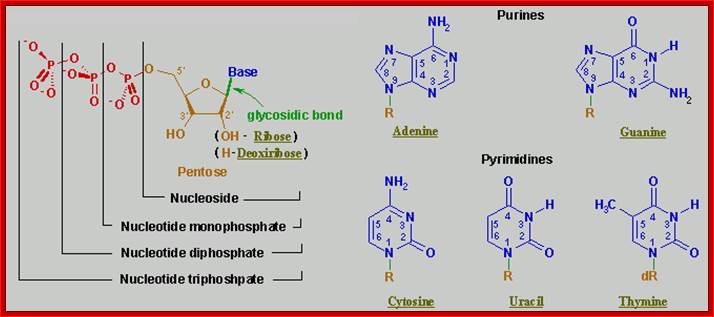
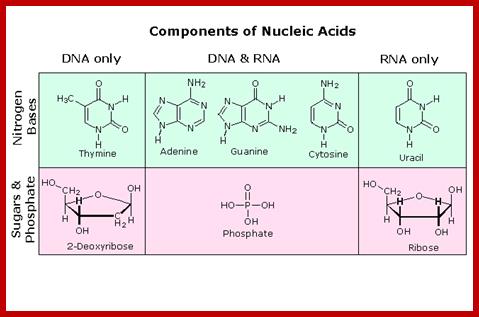
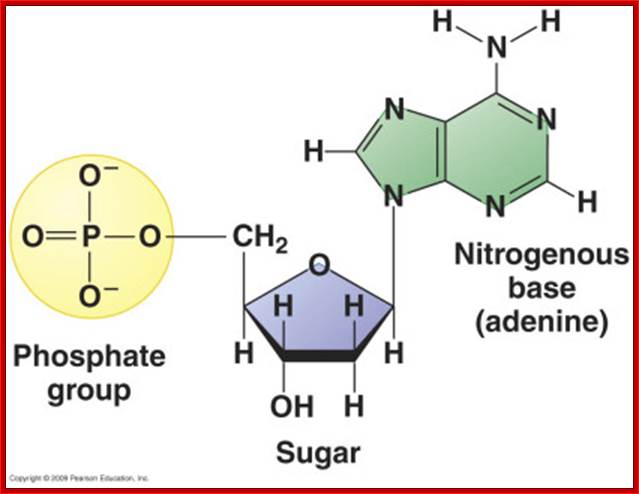
· DNA and RNA are composed of Phosphoric acid (H2PO4^ˉ2 nitrogenous base, and a pentose sugar.
· Pentose sugar is Deoxy ribose in DNA and Ribose in RNA. The Deoxyribose is lacking OH moiety at 2C’, and the ribose possess OH group at both 3C and 2C’ positions.
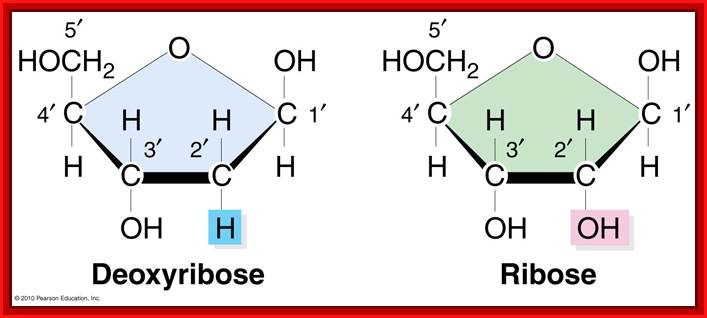
Pentose sugars- Deoxy Ribose an Ribose; look at positions of C in the ribose ring; http://twisteddnas.wordpress.com/
· The OH group at C-1 position is in beta configuration.
· Phosphate is phosphoric acid H2PO4^ˉ2, depends upon the ambient pH of the solution or the environment. It is phosphate group gives DNA and RNA acidic nature.
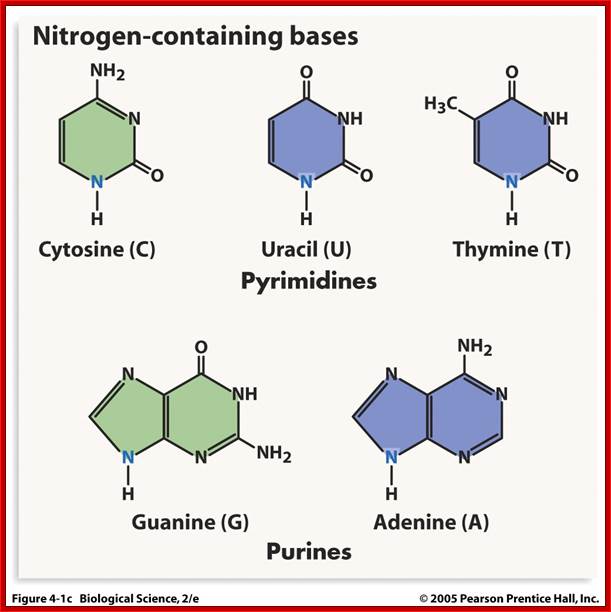
· Nitrogenous bases are of two types, one Purines and the other type is Pyrimidines.
https://www2.chemistry.msu.edu/
www.uic.edu
This is deoxyribose nucleotide with monophosphate; http://www.twisteddnas.wordpress.com
Numbering of atoms in rings:
Components of Nucleotides; Ribose pentose sugar is important for it is linked to Nitrogen base at its C’1 and three phosphates linked at 5’end of the sugar; Sugar in ribose both 2’ and 3’ have OH groups, but in Deoxyribose Oxygen is absent at 2’ of the sugar. Sugar with nitrogenous base and three phosphates at C5 is tri-pho nucleotides.www.en.wikipedia.org
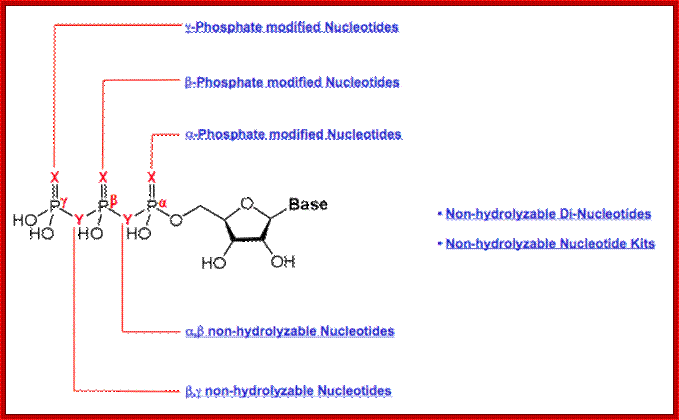
Nucleotide; look at positions of base and phosphates with respect to c’ positions of sugar. Also look at the positions of phosphates-alpha, beta and gamma positions. And look at phosphate with its P=O on one side and the other side it has P-OH group; http://www.jenabioscience.com/
|
Base Formula |
Base (X=H) |
Nucleoside X=ribose or deoxyribose |
Nucleotide X=ribose phosphate |
|
|
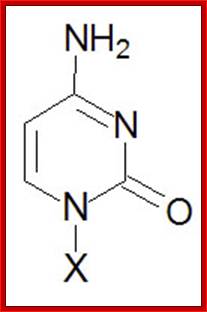
Cytosine, C |
Cytidine, C |
Cytidine monophosphate, CMP |
|
|
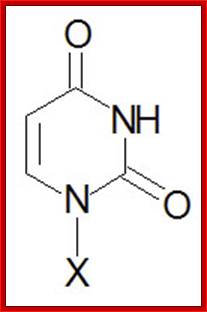
Uracil, U |
Uridine, U |
Uridine monophosphate, UMP |
|
|
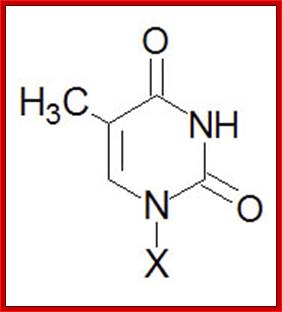
Thymine, T |
Thymidine, T (only Deoxyribose) |
Thymidine monophosphate, TMP |
|
|
Adenine, A |
Adenosine, A |
Adenosine monophosphate, AMP |
|
|
Guanine, G |
Guanosine, G |
Guanosine monophosphate, GMP |

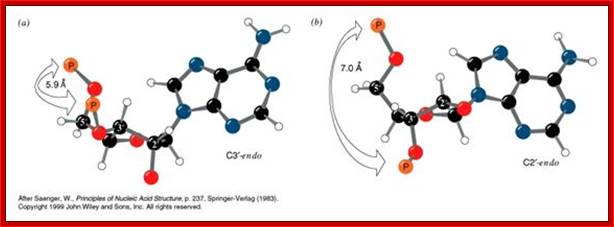
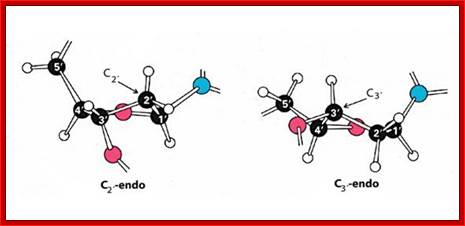
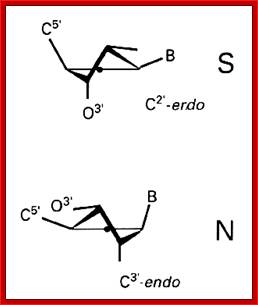
Structural features of nucleosides; with respect to orientation of N-bases one can call anti and syn; this orientation anti and syn is prominent when sugars go through their conformational changes such as C3 endo, C2 exo and vice versa; this is called sugar puckering; http://www.atdbio.com/ and www.scinceisart.com

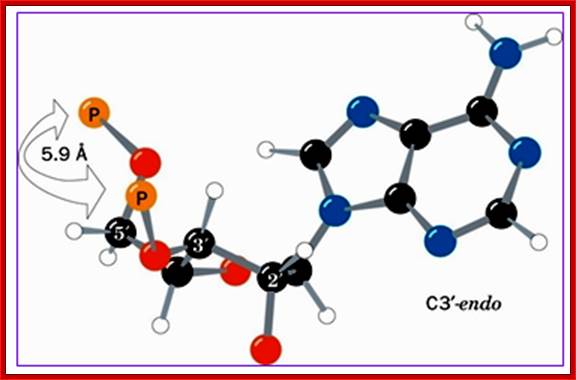
Nucleotide sugar conformations-Upper figure C3’ endo and C2’ exo, the lower figure contains C2’ endo and C3’ exo; this configuaration also change the orientation of the bas and sugar with respect to the carbon positions of the sugar. http://www.siumed.edu/
Purines, namely Adenine (A) and Guanine (G), each contain two heterocyclic rings with double bonds, and the bases are covalently linked to Ribose sugars at 1C’ by a glycosidic bond (between 9N’ of purine and 1C’ of pentose sugar), the resultant compound is called Nucleoside, which when added with a phosphate group to 5C’ of the sugar (i.e. CH2OH of the sugar), it forms an ester bond, and such a structure is called nucleotide.
Pyrimidines namely Cytosine(C), Thymine (T) and Uracil (U), each contain one heterocyclic ring with at least one double bond and they are linked to sugars through 1’N of pyrimidine with 1C’ of pentose sugar, which when added with a phosphate to 5’ it becomes pyrimidine nucleotide.
 Credit:Rahael Rettener, udaix Shutterstock; http://www.livescience.com/
Credit:Rahael Rettener, udaix Shutterstock; http://www.livescience.com/
Each nucleotide is composed of:
- A Sugar:
- Deoxyribose - a 5-carbon sugar in DNA
- Ribose - a 5-carbon sugar in RNA. This sugar has more oxygen than the sugar in DNA.
- One of these four nitrogen bases:
- Purines - have a double ring of carbon and nitrogen atoms.
- Adenine
- Guanine
- Pyrimidines - have a single ring of carbon and nitrogen atoms.
- Thymine
- Thymine is replaced by uracil in RNA.
- Cytosine
- A phosphate group - PO4
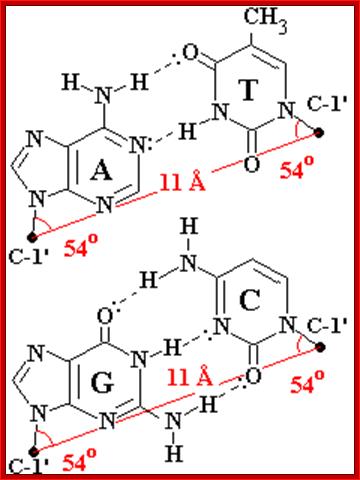
- Base pairing: The base pairing is always complementary between Guanine and Cytosine, and adenine and thymidine. This is called as Watson-Crick base pairing rule. There are deviations from this rule. But there are variations in base pairing; they are called Hoogsteen base pairing and reverse Hoogsteen and reverse Watson-Crick base pairing.
- What is exclusive of this specific base pairing? This rule stems from the fact that Guanine exists in ketonic form and cytosine in amino form in native state; similarly adenine in amino form and thymidine in ketonic form. This ionic form drive exclusive base pairing between G and C; and A and T. Otherwise any base could have base paired with any base. Dr. Donahue provided this ionic state information to J.D Watson.
· Characteristically, nucleotides exhibit specific conformation, which is very important in the 3D structural organization, stability and functions of DNA or RNA. In general, where the distribution of nucleotides in the chain is random, the sugar moiety shows a configuration of C2’ endo (puckered inwards to the plane of the ring) and C3’ exo (puckered out ward of the plane of the ring), or vice-versa, hence the 5’CH-O-phosphate group and the 1’C of the base are on the opposite sides of the sugar plane, so it is called anti-conformation, (because of sugar puckering) which is the most prevalent conformations in most of the ds DNAs.
Nucleic Acid Architecture: Tomado de: in a particular sequence in DNA the sugars undergo such conformational change that results in change in the structure of DNA.
http://www.chembio.uoguelph.ca/educmat/chm730/h730.htm
“anti- extends the base and pentose rings in opposite directions. For pyrimidines, this means that O-2 faces away from the pentose.
syn- orients the base and the pentose in the same direction”.
Ribose ring pucker arises because a flat pentagon puts all ring C atoms into eclipsed conformations, despite the fact that the pentagon angle of 108o is very close to the C tetrahedral angle of 109.5o. Puckering relieves the steric conflict due to eclipsed groups, at the expense of a slight deviation from tetrahedral bond angle. The pucker is described in terms of the pentose ring atom that is displaced furthest out of plane. When an adjacent atom is slightly displaced to the opposite side, this gives the 'S' or 'N' pattern twists, according to the orientation of the zigzag.
Christoph Schneider, -- Biocomputing Group ;http://jenalib.fli-leibniz.de/
Structural features of nucleosides
The X
distribution of the diffraction pattern indicates 2-fold symmetry; hence
an antiparallel pair of strands was used in the model.
The
X-ray diffraction pattern provided by Miss. R. Franklin and Dr. Williams showed
antipolarity of the helical strands and ionic phosphate groups are excluded
from the center of the helix and found at the periphery of the helix. Did
Watson and Crick give credit to Franklins’ work? In a race to Nobel laureate
title Linus Pauling and Franklin missed (Miss Franklin’s demise early robbed
her credit to the discovery to intelligent ds DNA model).
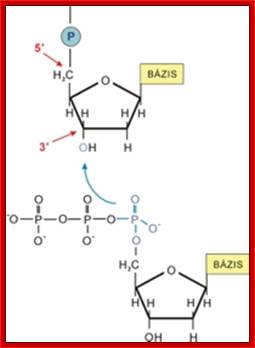

Joining of two nucleotides to dform a dinucleotide chain; www.uic.edu
www.ibhumanbiochemistry.wikispaces.com
web2.mendelu.cz

Watson-Crick-Base Pairing; http://tonga.usp.edu/
Molecular Dimensions; http://tonga.usp.edu/
This general rule, however doesn’t apply under certain situation where the nucleotide sequence is 5’CG CG CG CG 3’ and its repeats. In such cases, if the sequence is repeated and extends to 6 or more than six base pairs in a stretch, the purines especially Guanines take C2’exo, and C3’endo, this leads to cis configuration, that is both 5’CH2O-P group and the base at1’C are towards the same side of the plane of the sugar, because of one purine and one pyrimidine alternating to each other in a stretch of six or more base pairs force the sugar to such puckering, which renders the sugar in to stressed state. On the contrary the pyrimidines configuration remains ‘anti’ conformation. Such sequences (CG) n or (GC)n are localized in certain segments of the DNA; their position and the size varies from one species to the other. Methylation of C s in CG repeats produces certain ‘syn’ configuration of G.
All single stranded DNA and RNA molecules have nucleotides with anti configuration with respect to their sugar puckering conformation. Nucleotides, because of their heterocyclic rings and double bonds, strongly absorb light at UV region I.E 250-280 nm. As they possess heterocyclic rings with double bonds they show hydrophobic features.

https://chelseaharripersad.wordpress.com
|
Parameters |
A Form |
B Form |
Z-Form |
|
Direction of helical rotation |
Right |
Right |
Left |
|
Residues per turn of helix |
11 |
10 |
12 base pairs |
|
Rotation of helix per residue (in degrees) |
33 |
36 |
-30 |
|
Base tilt relative to helix axis (in degrees) |
20 |
6 |
7 |
|
Major groove |
narrow and deep |
wide and deep |
Flat |
|
Minor groove |
wide and shallow |
narrow and deep |
narrow and deep |
|
Orientation of N-glycosidic bond |
anti |
anti |
anti for Pyrimidines, syn for Purines |
|
Comments |
|
most prevalent within cells |
occurs in stretches of alternating purine-pyrimidine base pairs |
RNA polynucleotide chain is formed by triphopho- ribonucleotides join to one another, but require a template it can be RNA or DNA.
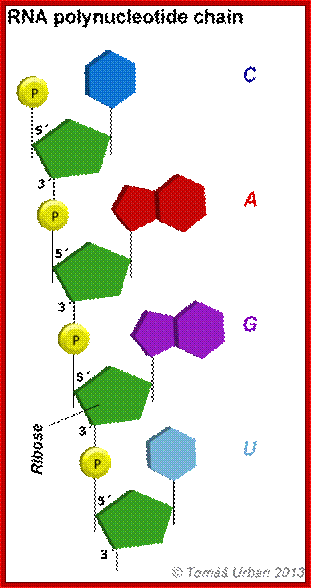


Primary structure of RNA; At the 5’end it has 5’P-OH and 3’end it has 3’OH http://chemistry.tutorvista.com