Cellular Signal Transduction IV-
Second messengers and Receptor-Transporters:
Signal transduction often generate second messengers such as 3’5-cyclicAMP, 3’5-cGMP, cyclic ADB-ribose (cADPR), 1’2-Diacyl glycerol, 1’,4,5-Inositol triphosphate, Nitrc oxide and calcium ions. Proteins that bind to second messengers are called switch proteins. Many hormones cause elevated levels of second messengers which activate cellular enzymes and involved in cellular metabolism-protein kinases and phosphatases.
http://www.rpi.edu

http://www.epo.wikitrans.net; top-fig-Phosphatidyl Inositol 4,5 phosphate, 1,3 phosphate

https://www.thinglink.com

https://www.rpi.edu/

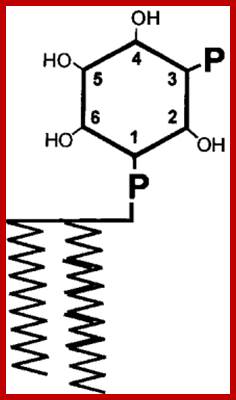
Inositol 1,2,3,4,5,6-phosphate (a 6-carbon Inositol); https://www.clearsynth.com
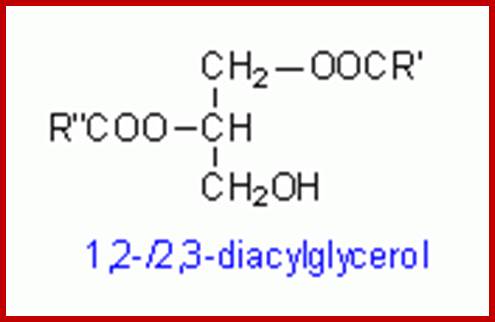
Diacyl glycerol:
http://www.lipidhome.co.uk
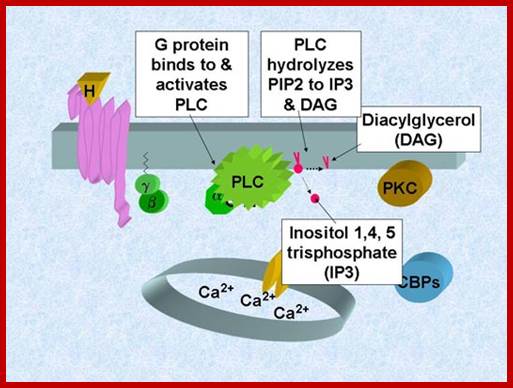
The DIAG are membrane bound yet their action is pervasive. This membrane bound second messenger is generated when GPCR activated G-alpha induced Phospho lipace-C cleaves Phophotidylinositol4, 5-phosphate to release inositol1, 4, 5-inositol and DIAGDIAGs interact with a family of receptors. Signal transduction pathway by G protein pathway generates DIAG and PI3.
The DIAG activates PK-C which in turn activates MAPK pathway and increase Ca responsiveness of contractile elements. Through MAPK pathway it can activate few genes.
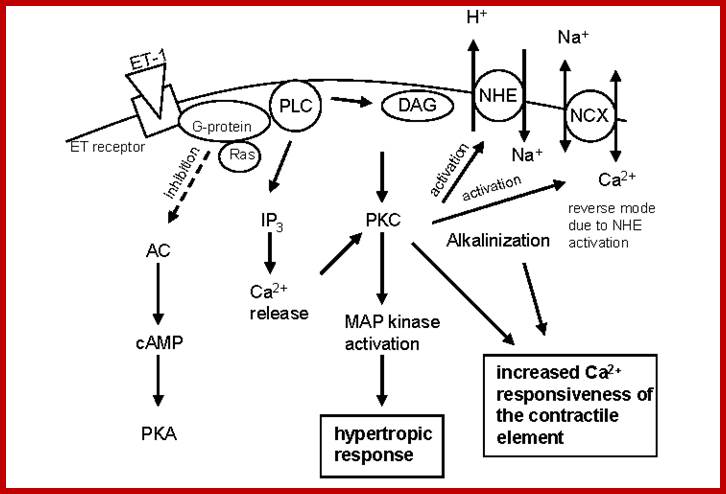
http://www.lipidhome.co.uk
T cell Receptor activation:
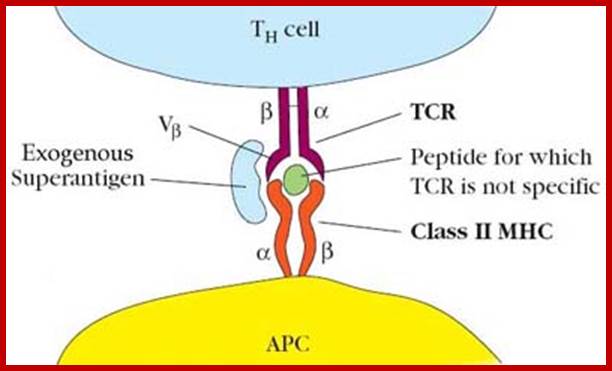
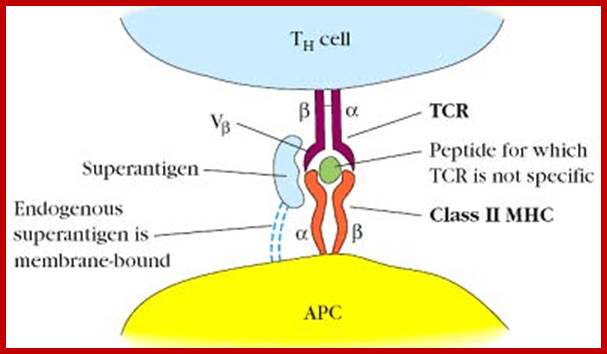
Exogenous super antigens; http://biosiva.50webs.org/
T cell independent antigen – I:

It does not require T cell to induce immune response. http://biosiva.50webs.org
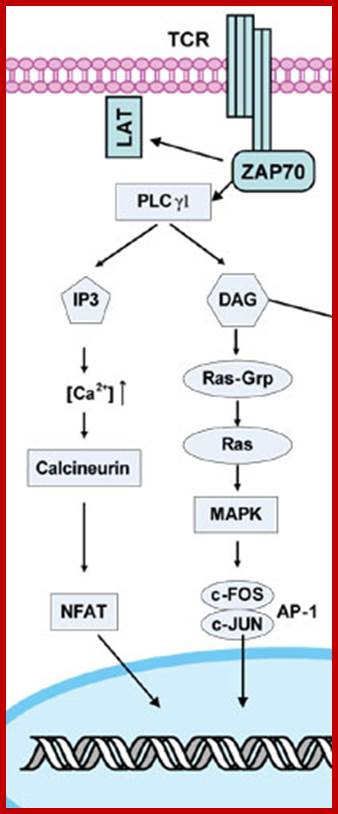
Ability of an antigen to induce immune response is known as antigenicity. Ability of immunogens to induce immune response is known as immunogenicity. These two terms used interchangeably.;T-Cell Receptor activation
G-protein mediated transduction; www.utm.utoronto.ca/phorbol

https://www.slideshare.net/
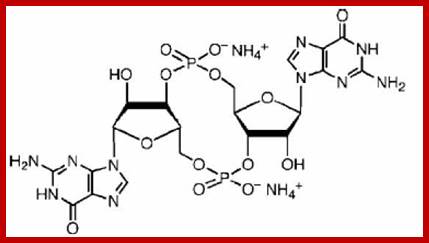
cyclic ADP Ribose
Di G 3-5, 5-3diphosphate; http://jb.asm.org
Cyclic ADP ribose:
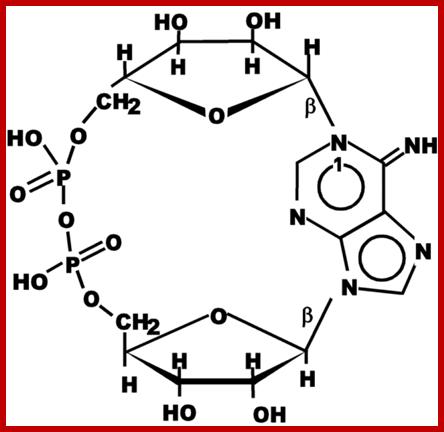
Cyclic ADP ribose http://ajplung.physiology.org
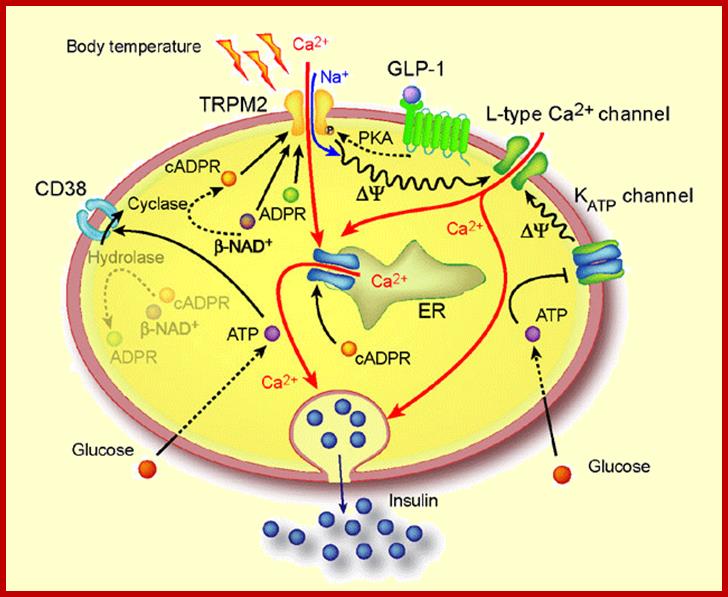
ADPCR: This molecule has multifarious functions such as activating and the releasing of Ca+ ions from ER, opening of PM Ca+ channels for the entry of ions into cells, activating Ca/Na channels, and activates release of insulin granules into blood stream.
https://www.slideshare.net
Ca/Na Mediated release of Insulin granules
Nitric oxide (NO):
Nitric oxide in smooth muscle cells causes cGMP level to be increased by guanylyl cyclase activity. No is synthesized from Arginine conversion to citrulline by NO synthase. Endothelial cells lining blood vessels are activated by Nitric oxide synthase. Acetyl choline acts on GPCR which acts on PLC which releases IP3 which activates the release of Ca. The Cal-D in turn acts on Nitric oxide synthase. This enzyme acts on Arginine to generate No and citrulline. The NO acts on smooth muscle cell NO receptors and activate the receptors to activate the synthesis of cG-cyclase that generates cGMP. The cGMP binds to protein kinase G (PKG). The PKG in turn activates signaling pathway leading to the inhibition of Actin-Myosin complex formation, thus makes the smooth muscles relaxed around blood vessels. Penegra and Viagra are used for penile errection deficient people, often is used in curing heart attack to dilate blood vessels.
Arginine + O2Citrulline-> NO,
The No diffuses through smooth muscle cells and causes increase in cGMP which in turn cause smooth muscles to relax; in fact nitroglycerine is used in treating heart muscles to relax.
Nitric oxide is released by the activity of Nitric oxide synthase through the use of Arginine as the substrate. The intra cellular NO activates cyclic guanylyl cyclase releasing cGMP, which in turn activates protein kinase-C, which has far reaching effects such as relaxation of smooth muscle cells; important in arterial blood vessels, inhibits platelets and changes gene expression.
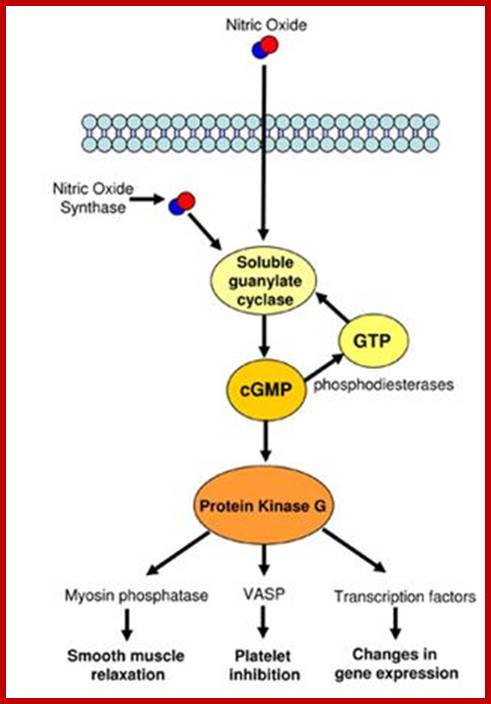
Nitric oxide as signal molecules; www.reading.ac.uk/cellmigration; NO induced activity; ;http://www.reading.ac.uk

https://www.researchgate.net
Effects of cAMP:
Release of cAMP via GPCR and adenyl cyclase has far reaching effects in cytosol, but one of the important action is to activate protein kinase-A, which has a cascading effect.
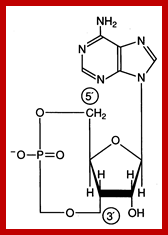
cAMP; https://www.studyblue.com
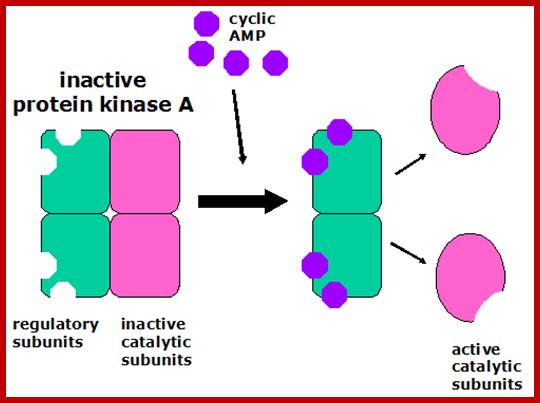
https://www.studyblue.com
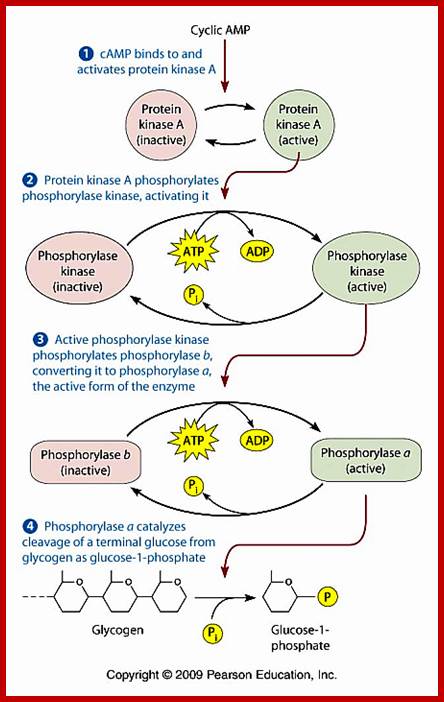
http://www.mun.ca/
Effect of cGMP:
Signal induced activation guanylyl cyclase acts on GTP and converts into cGMP. This second messenger has various impacts such as activation of cGMP dependent protein kinase 1, cGMP activated Ca/Na co transporters, activates cAMP dependent phosphodiesterase and also works through nitric oxide in relaxing smooth muscles.
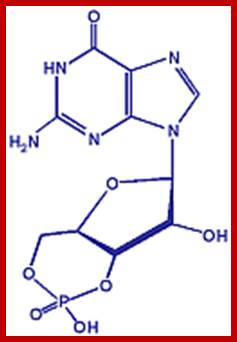
cGMP

https://www.slideshare.net
:
www.slideplayer.com/
cAMP, cGMP, ADRPP, NO and calcium ions play an important role as second messengers.
cAMP :
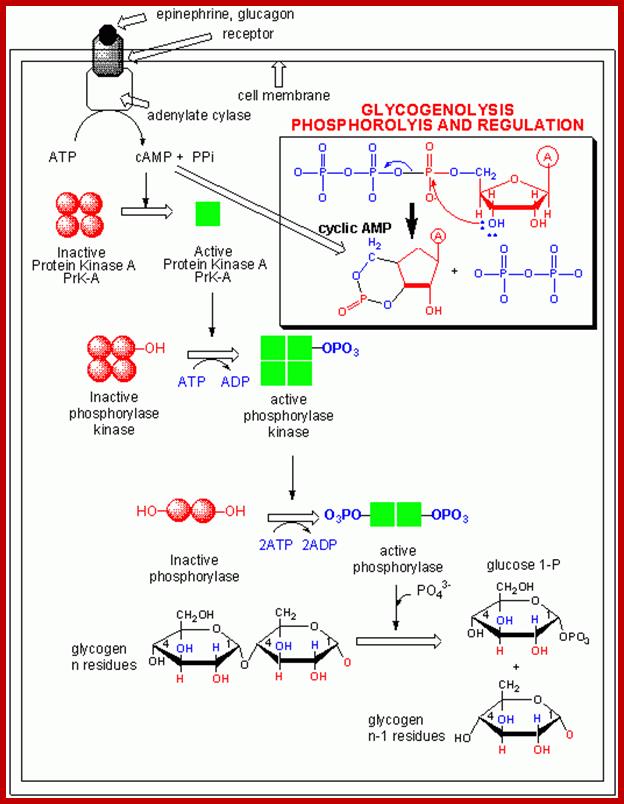
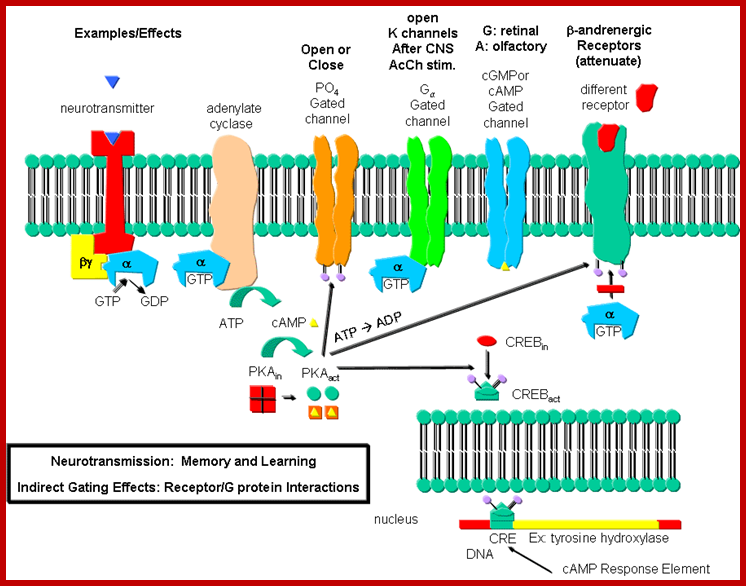
cAMP activates protein kinase A ( PKA). PKA in cells remain inactive by an inhibitor. Cyclic AMP binding to the inhibitors frees PKA as an active form. PKA phosphorylate glycogen phosphorylase kinase; this enzyme in turn phosphorylates glycogen phosphorylase which leads to the breakdown of glycogen into glucose1-phosphate. cAMP dependent PKA also phosphorylates cyclic Amp response element-binding protein CREBP, which acts as TF and binds to CRE (cAMP response element) and activate specific genes. cAMP also interacts with cAMP gated cat ion channels of olfactory receptors in neurons resulting in Na ion influx and membrane depolarization.

Effect of cAMP on immune, tumor, and epithelial cells. Impact and function of cyclic adenosin monophopshate (cAMP) on T and B lymphocytes, granulocytes, monocytes, macrophages, dendritic cells, epithelial cells, and melanoma cells. LTB4, leukotriene B4; LTC4, leukotriene C4. The cAMP Pathway as Therapeutic Target in Autoimmune and Inflammatory Diseases; Verena Katharina Raker et al; Mainz et al Germany; http://journal.frontiersin.org/
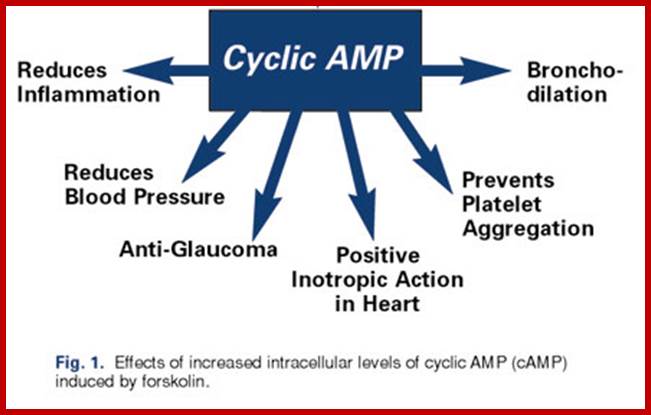
Forskolin and cAMP – Wide-Ranging Benefits From Ayurvedic Herb
James English; https://nutritionreview.org
All the effect of cAMP is executed through PKA for it is cAMP dependent kinase. PKA perse is made up of 4 subunits, 2regulatory units and 2 catalytic subunits. In unbound form the catalytic subunits are sequestered. When cAMP binds subunits of the complex undergo conformational change releasing subunit active form.
In adipose tissue epinephrine induced PKA promotes p-Lation of phospholipase which catalyzes hydrolysis of stored triglycerides to yield free fatty acids and glycerol. These are released into blood metabolized to generate energy.
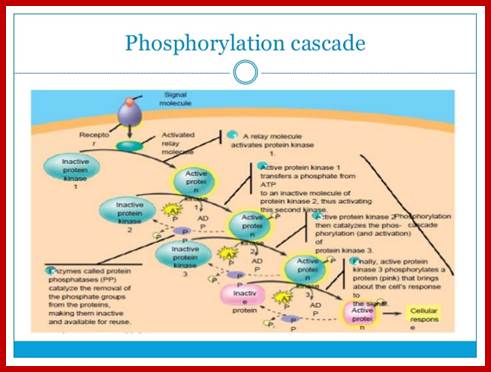
PKA , whatever may be the substrate , it p-lates serine/threonine with a sequence such as YTRCR/K. Glycogen is metabolized by cAMP induced PKA in a cascade manner. As the requirement of cAMP is very high initial production of cAMP gets amplified through receptor activation. A single receptor with the binding of the ligand can convert inactive Gs-alpha into active form. And each Gs-alpha activates the synthesis of several hundreds of cAMPs before the ligand dissociates. Such amplifications occur with other G-coupled proteins involved in second messenger synthesis.
https://www.slideshare.net/

Regulation of glycogen metabolism; https://www.studyblue.com
To cite that a single epinephrine binding triggers a cascade of diverse reactions resulting in the production energy source available to the body. Termination of GPCR mediated reactions performed by beta adrenergic receptors and regulated by several other factors. Sensitization of the receptors for long time leads to desensitization.
www.slideplayer.com/
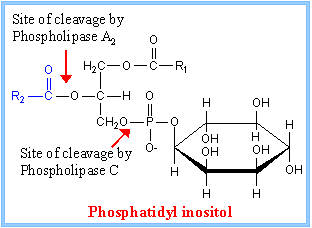
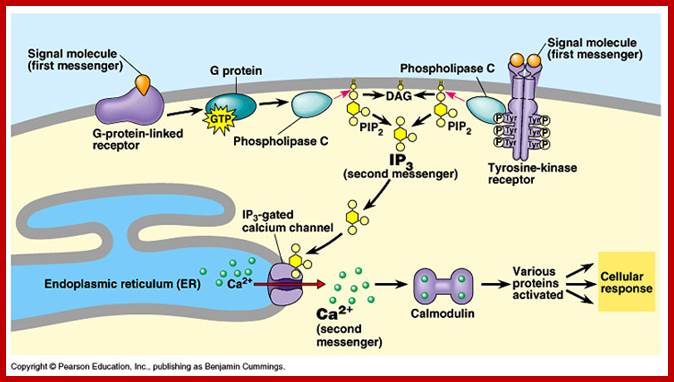
Phosphatidyl Inositol generates many second messengers:
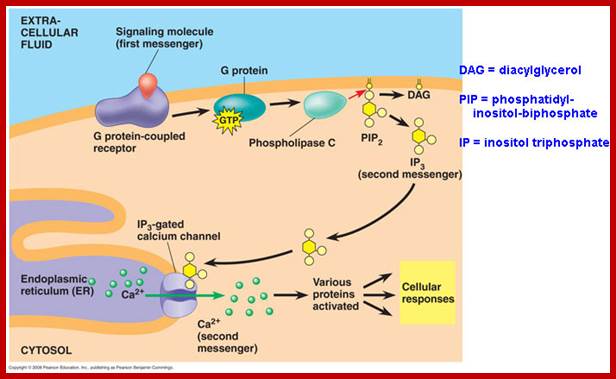
Besides cAMP production many other second messengers are produced in response to specific hormones by activating GPCR. Several hormones binding to Go or Gq receptors induce activation of Phospholipase-C beta. PLC beta is an effector in this case. Phospholipase-C generates second messengers like 1’2’-DIAG (remians associated with membrane) and IP3.
PI3, Inositol1’4’5’-P triggers the release of ca ions from ER which is the repository Ca ions, even mitochondrial act as the repository of Ca ions. Cytosolic Ca level 0.2um is always maintained, otherwise higher levels can trigger a variety of responses, hence its level is controlled. Binding of hormone induce the elevation of cytosolic Ca by IP3 mediated and gated Ca channels. The channel proteins are made up of 4 identical subunits and each contain IP3 binding sites. Ip3 binding induces conformation changes in the transport proteins and open. Its release is temporary and soon they are pumped to exoplasmic space or ER or both Ca-ATPase pumps. Higher conc. of calcium decreases the affinity of IP3 to the transport proteins.
https://www.slideshare.net
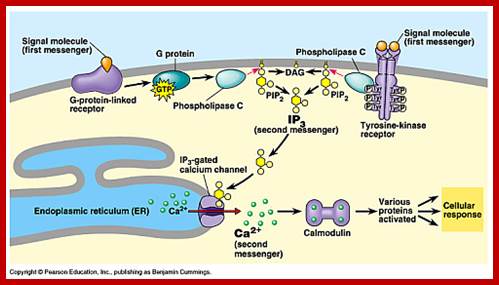
Calcium ions as second messengers act as signals in a wide variety of cellular activities. In animal cells transient increase in Ca2+ ions in response to certain hormones is due to the release of them from intracellular compartments rather than from extracellular regions. However hormone induced opening of calcium channels is governed by another second messenger called Inositol triphosphate ((IP3); it is a minor component of membranes. The PI can be converted to polyphosphoinositides PI phosphate (PIP) and PI biphosphate (PIP2) by kinase activities. In animal cells binding of a hormone such as Vasopressin to it receptor leads to the activation of trimeric G proteins. The alpha subunit dissociates and activates Phospholipase C (PLC). The activated PLC hydrolyses PIP2 generating IP3 and DIAG.
IP3, a water soluble compound diffuses in cytoplasm and binds to vacuolar tonoplasts; they are IP3 gated Ca ion channels and open.
Cyclic ADP ribose cADPR another kind of second messengers and they can release Ca ions from the internal stores. While cAMP activates PKA, cADPR activate specific Ca+ ion channels.

http://slideplayer.com/
slideplayer.com/slide
GPCR with specific ligand binding activate PLC isoform leading to IP3 mediated increase in cytosolic Ca in specific cell types. Acetyl choline induced GPCR ends in Ip3 mediated rise in Ca that triggers secretory vesicles with PM to release their contents into extracellular space. A rise in Ca in blood platelets induced by thrombin stimulates cell fragments aggregate and plug holes in blood vessels. Secretion of insulin from beta cells is also triggered by Ca+ of course by another mechanism. Calcium ions can activate a host of cellular responses.
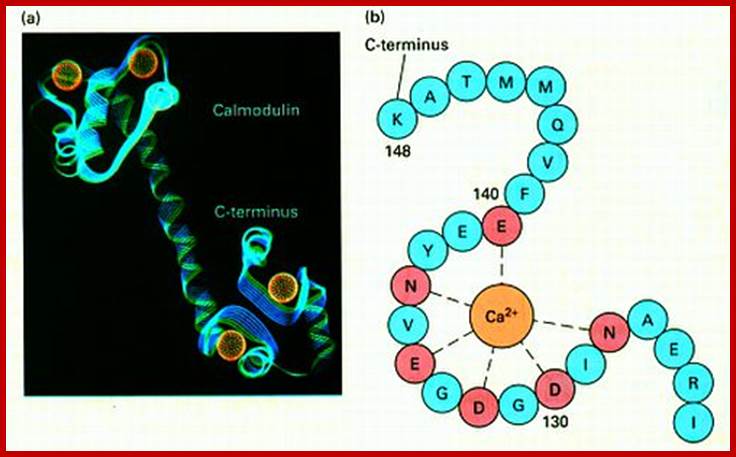
Calmodulin:
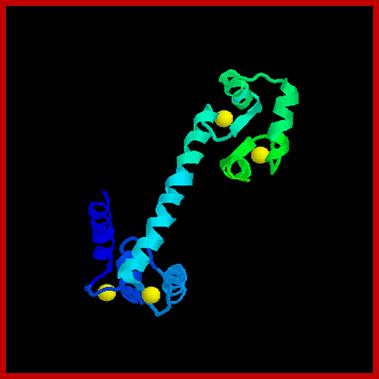
Cellular responses by Ca+ are achieved by the binding of calcium to a protein called calcium binding protein called Calmodulin. Each Calmodulin protein binds to four Ca ions and changes conformation enabling it to bind and activate several proteins such as Ca-ATPase. Calmodulin is ubiquitous in cells and act as switch protein that mediates many cellular effects of ca ions. The activity is brought about by the activation of Ca-Calmodulin-dependent protein kinase (CaM). The CAM is serine/threonine kinase cause enzyme activation. The CaM perse consists of two globular ends separated by flexible helix.
http://www.whozoo.org/
www.cc.scu.edu.cn/G2S/Template/
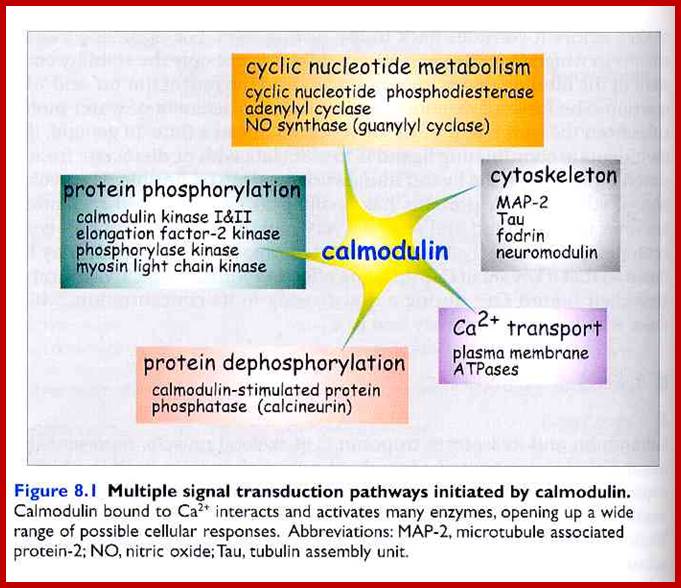
Each globular ends contain two Ca+ binding sites. When CAM binds to proteins it actually wraps around it. Myosin light chain kinase in muscles is activated by Calmodulin. Similarly cAMP phosphodiesterase (cAMP-pDE) is activated by Cal-D . In some, Ca-D activates certain protein kinases which p-lates certain TFs thus regulate gene expression. In T-cells Ca ions enhance the activity of an essential TF called NAFT (Nuclear Factor activated T cell). In unstimulated cell, NAFT remains in cytosol, but activated receptor mediated increase in Ca, Ca-D binds to Calneurin (a protein phosphotase) and activates it. The activated Calneurin dephosphorylates cytosolic NAFAT thus it moves into the nucleus and stimulate the expression of genes for activation of T-cells. Cal-D plays a role in dilation of blood vessels to deliver oxygen. Calmodulin is involved in cyclic nucleotide metabolism, protein phosphorylation, and activation of cytoskeleton proteins, protein dephosphorylation and Ca+ transport.
Diacyl glycerol activates protein kinase C:
Cleavage of PIP2 by phospholipase C releases DIG and IP3. DIAG remain associated with membranes activates a family of Protein kinases collectively called Protein kinase-C (PKC). The PKC in turn associates with PKC receptor proteins. Phospholipase A2 and Phospholipase-D generate other membrane derived signals. There is a connection between IP3 and DAG. Released Inositol phosphates induce the channel proteins found in ER to release Ca ions.
Activation of PKC induces a variety of cellular responses such as cell growth and metabolism. In liver PKC facilitates in regulating glycogen metabolism. PKC also p-lates several TFs and induce differential transcription.
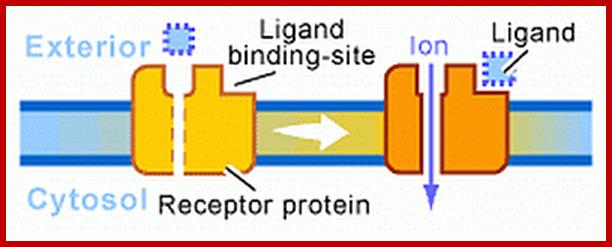
Receptor linked Ion channels:
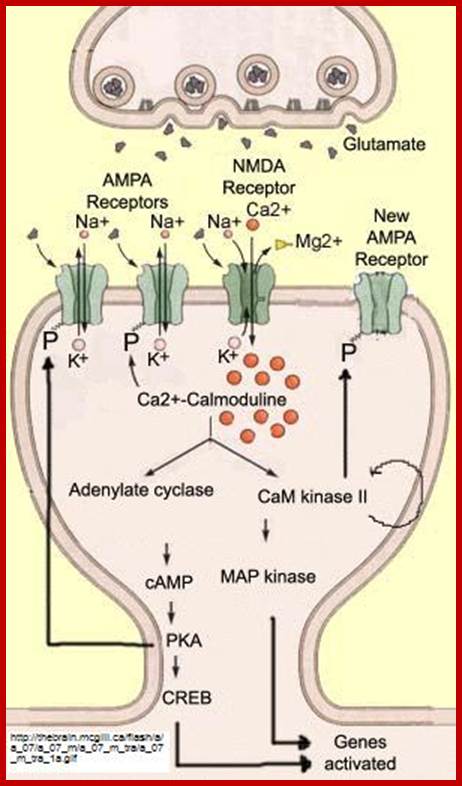
There are innumerable ion channels used in transport of a variety of ions both metal ions and nonmetal ionic and nonmetal ionic channel proteins. But there are some important transporters that act as receptor Ex. Glutamate receptor, calcium ion channels.
www.slideplayer.info/slide
https://employees.csbsju.edu

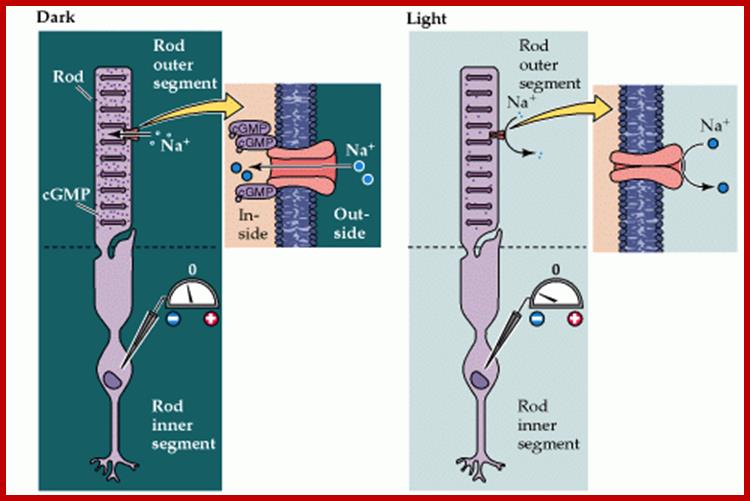
Light induced cGMP mediated Na channel opening and closing
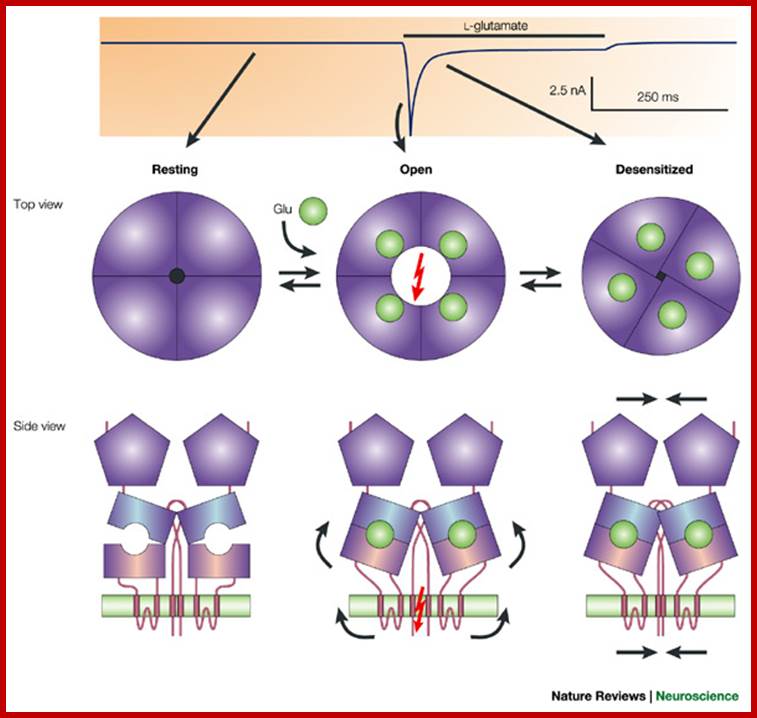
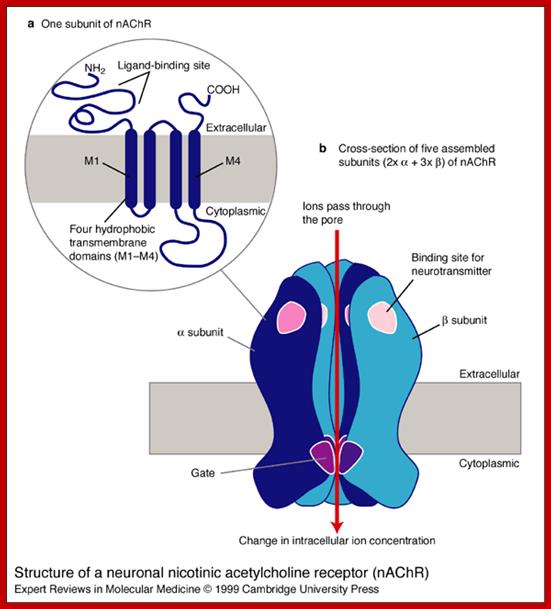
Structure of a neuronal nicotinic acetylcholine receptor (nAChR). The nAChR is a ligand-gated ion channel. Each nAChR is composed of five (a or b) subunits, which can be homomeric (i.e. all a or all b) or heteromeric (i.e. a mixture of a and b, as in this schematic). (a) One subunit of the nAChR comprises: (1) an N-terminal extracellular domain, which is involved in binding to (neurotransmitter) ligands, (2) four hydrophobic transmembrane domains (in dark blue), called M1, M2, M3 and M4, and (3) a long cytoplasmic loop between M3 and M4, and other shorter loops connecting the domains. (b) In cross-section, an assembled nAChR has five subunits, each with a binding site and a gate region. All ligand-gated ion channels bind specific neurotransmitters (in this case, nicotine), which induce a conformational change in the receptor, opening the channel. Charged amino acids line the channel pore and select the ions that can pass through into the cell. The a4 subunit of the nAChR is encoded by the CHRNA4 gene, which is one of the genes that are mutated in autosomal dominant nocturnal frontal lobe epilepsy (ADNFLE).;Louise Bate and Mark Gardiner;http://journals.cambridge.org